From Startup Chaos to Scalable Success: The Big-League Playbook for Small Business Excellence
Stop fighting daily fires and start engineering your company's future with confidence. Our latest article reveals the blueprint for wiring your organization for lasting excellence and sustainable growth.
From Rigid Roadmaps to Agile Realities: Transforming Your Med-Tech Software
Start building safer, more valuable medical software while minimizing waste and meeting compliance with confidence. Read our post to learn how to transform your development process from a rigid roadmap to an agile reality.
The EHDS: A New Era for Health Data—Exciting and Scary
The EHDS will force the sharing of proprietary health data, directly challenging your company's market position and IP. Understand the risks and learn how to prepare your business for this new era by reading our essential analysis.
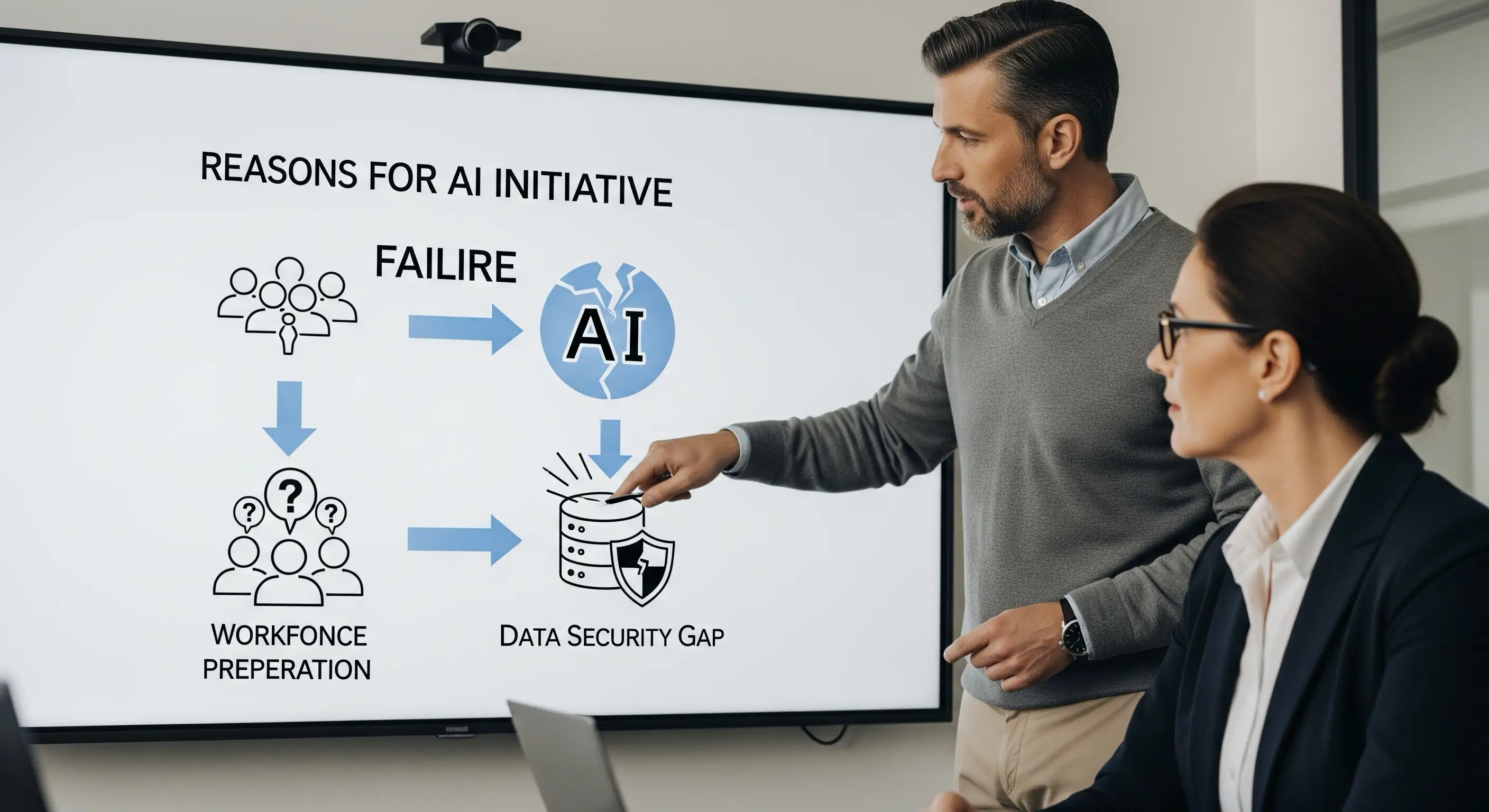
Beyond the Hype: Why 80% of AI Projects Fail at the Data Laye
Ready to move your AI project beyond the proof-of-concept graveyard and into production? Secure your data layer with our expert guidance and build the trustworthy foundation your AI needs to deliver real business value.
Building Unstoppable Teams: From Dysfunction to High-Performance
A cohesive, high-performing team is your ultimate competitive advantage, but it doesn't happen by accident. Learn how our structured approach transforms team dynamics and drives collective results by exploring our Managerial & Leadership Effectiveness service.
The Shadow Side of AI: Understanding Malicious Use and the Governance Imperative
Don't let the risk of malicious use derail your company's AI initiatives or expose you to unacceptable risk. Our latest article explains how to implement a proactive AI Governance framework that protects your business and enables safe innovation.
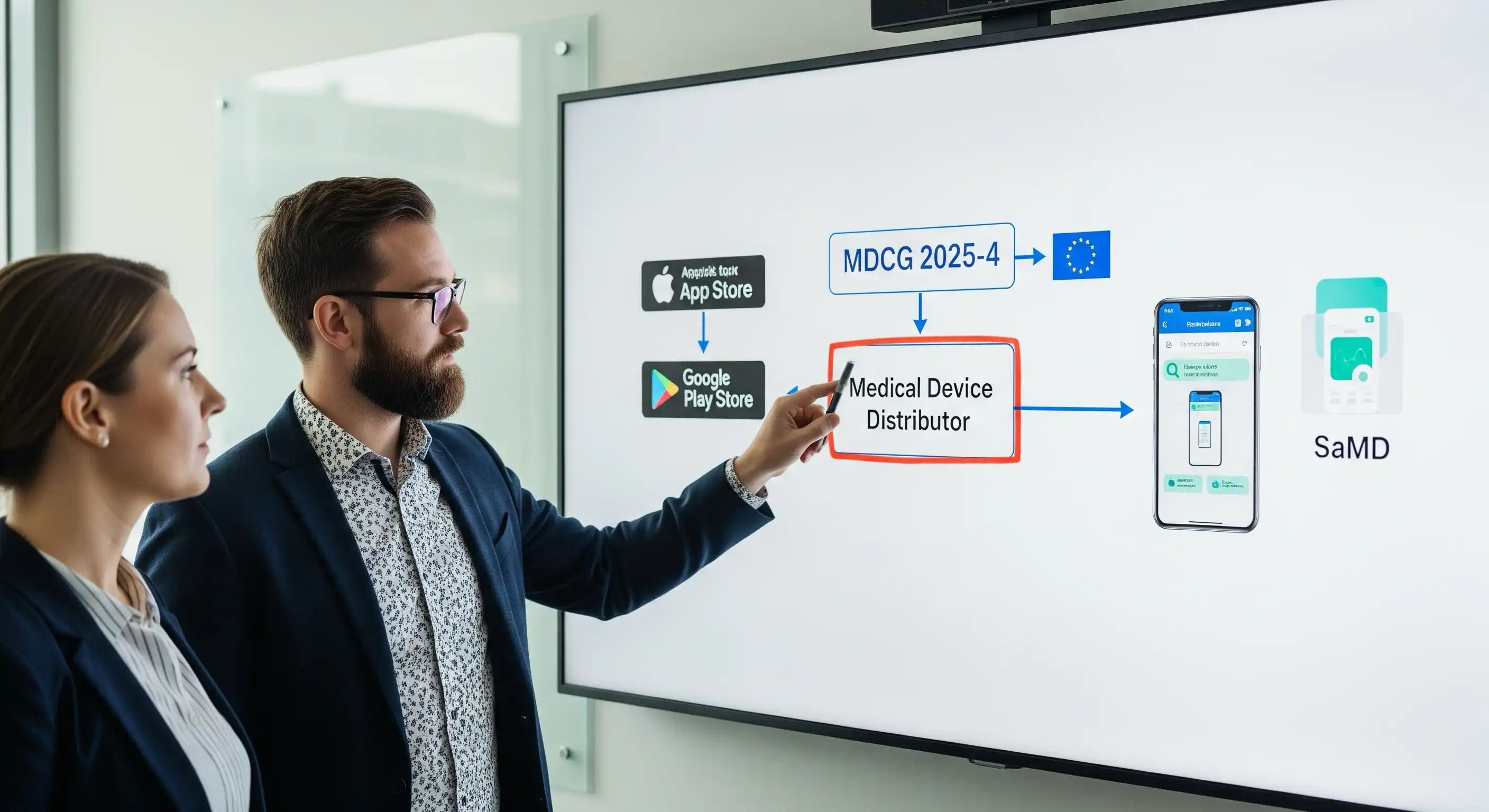
A deep dive into the landmark MDCG 2025-4 guidance and its impact on app stores and digital health developers.
With app stores now sharing regulatory accountability, the path to market for SaMD and digital therapeutics has never been clearer. Discover how this new era of shared responsibility benefits developers and strengthens the entire digital health ecosystem in our latest post.
A strategic framework for leaders to balance optimizing today's business while systematically building the company of tomorrow.
True strategic innovation requires balancing today's profits with tomorrow's growth opportunities. Learn how to manage your innovation portfolio across three distinct horizons and build a resilient future by design, not by default.
An analysis of the MDCG 2019-11 Rev. 1 guidance update. Discover the significant changes for Medical Device Software (MDSW), AI, modular software, and EHR systems under the EU MDR.
The EU's regulatory focus on AI and modular software is intensifying, and ambiguity in your intended purpose is no longer acceptable. Stay ahead of the curve by reading our breakdown of the critical changes in MDCG 2019-11 Rev. 1.
Building Digital Trust: A Guide to the ASME V&V 40 Standard for Medical Device Modeling
Don't risk a regulatory rejection because the credibility of your computational model is questioned. Our guide to the ASME V&V 40 standard provides the risk-informed framework you need to build irrefutable digital trust and streamline your submissions.
Audio Overview: Verification and Validation Activities for Computational Models with ASME V&V 40-2018
Computational modeling is reshaping regulatory pathways, with the FDA now recognizing digital evidence as a key part of device submissions. Press play to hear our deep dive into the ASME V&V 40 standard and learn how to build the model credibility required to accelerate innovation and de-risk your regulatory strategy.
EU's New AI Guidance for MedTech (MDCG 2025-6): A Welcome Map with Missing Roads
With Notified Bodies now assessing AI Act requirements alongside MDR/IVDR, your QMS and technical documentation must be fully integrated. Use our expert breakdown of MDCG 2025-6 to navigate this dual compliance landscape and position your AI medical device for success.
Audio Deep Dive: MDCG 2025-6 Explained - Navigating AI, MDR, and IVDR
Navigating the intersection of the EU AI Act, MDR, and IVDR is the biggest regulatory challenge facing Medical Device AI developers today. Press play for our expert audio deep dive, explaining the new MDCG 2025-6 guidance and providing the clarity you need to build a compliant strategy.
Beyond Consent: How French Guidance is Reshaping AI Development in the EU
France's CNIL has created a critical blueprint that will likely influence how AI is regulated across the EU, offering a path beyond consent-only data models. Our analysis explains the three key conditions you must master to leverage this new flexibility and innovate responsibly.
From Wish Lists to Roadmaps: The Art of Crafting Effective User Stories
A well-managed product backlog is your single source of truth for delivering value, and effective user stories are its foundation. Read our comprehensive guide to master the art of crafting, planning, and splitting user stories to transform your backlog from a wish list into a powerful roadmap.
Anatomy of a Turnaround: Rewiring a MedTech Company for Excellence
Is your company suffering from enterprise inertia, broken processes, and a culture that stifles innovation? Our case study provides a deep diagnosis of these systemic dysfunctions and reveals the multi-layered strategy required to rewire a MedTech organization for sustainable excellence.
Decoding Corporate DNA: A Cultural Analysis of MedInnovate Corp.
Generic change initiatives often fail because they ignore the invisible force of company culture. Discover how we use cultural analysis to design a precise, evidence-based roadmap that counteracts specific dysfunctions and rewires an organization for sustainable excellence.
Navigating the New Frontier: Key Changes in the FDA's 2025 Medical Device Cybersecurity Guidance
With the introduction of the "cyber device" and legally-binding requirements, manufacturers can no longer treat cybersecurity as an afterthought. Our latest article unpacks the critical updates in the FDA's 2025 guidance so you can adapt your quality system and protect your products.
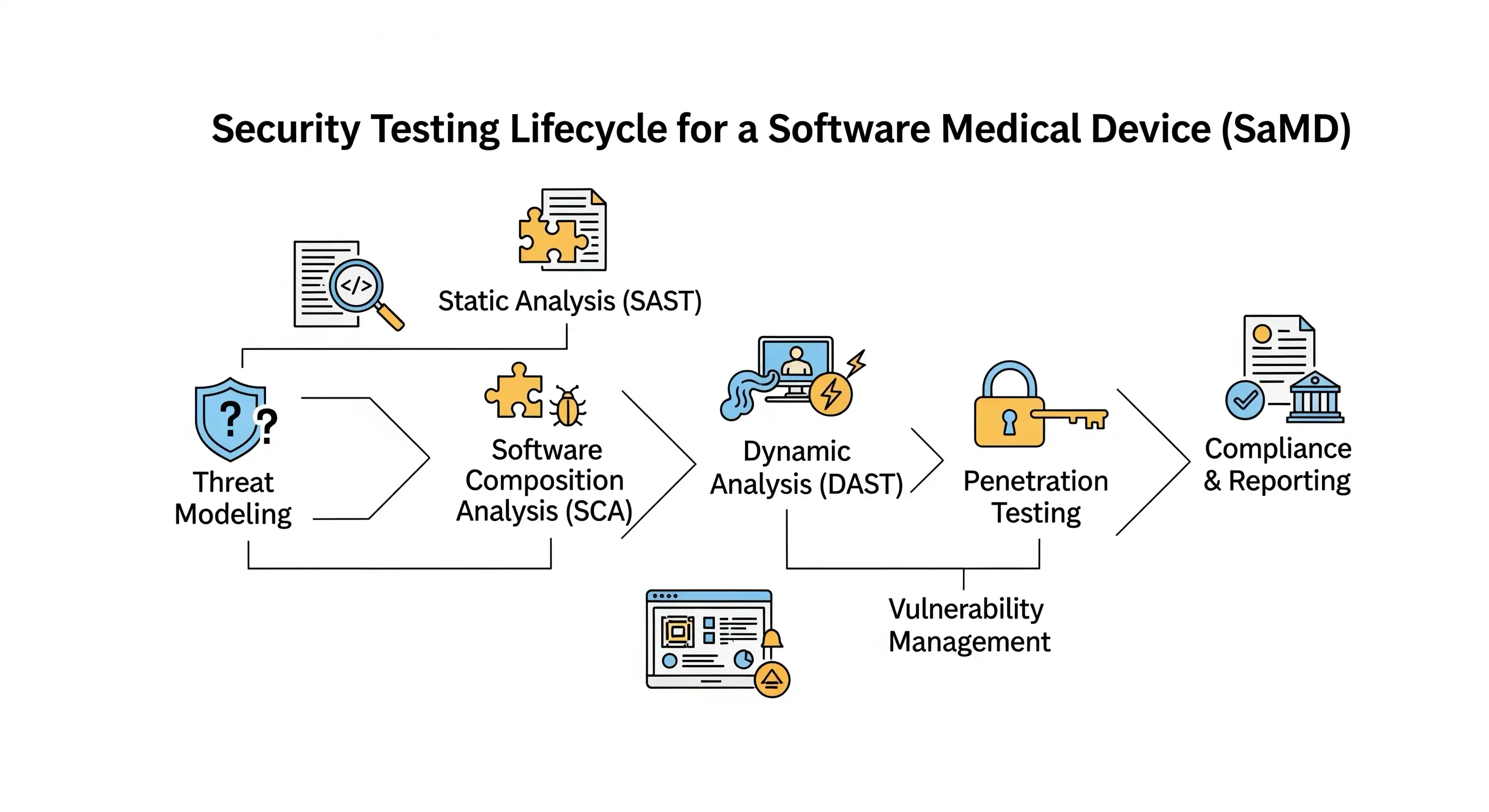
FDA's 2025 Cybersecurity Guidance: Mastering the New Testing Requirements
The FDA's new cybersecurity guidance is no longer a set of recommendations; it's a legal mandate with significant new requirements for all "cyber devices." Our in-depth article breaks down the critical changes and provides a detailed walkthrough of the four pillars of testing you must now master for your premarket submission.
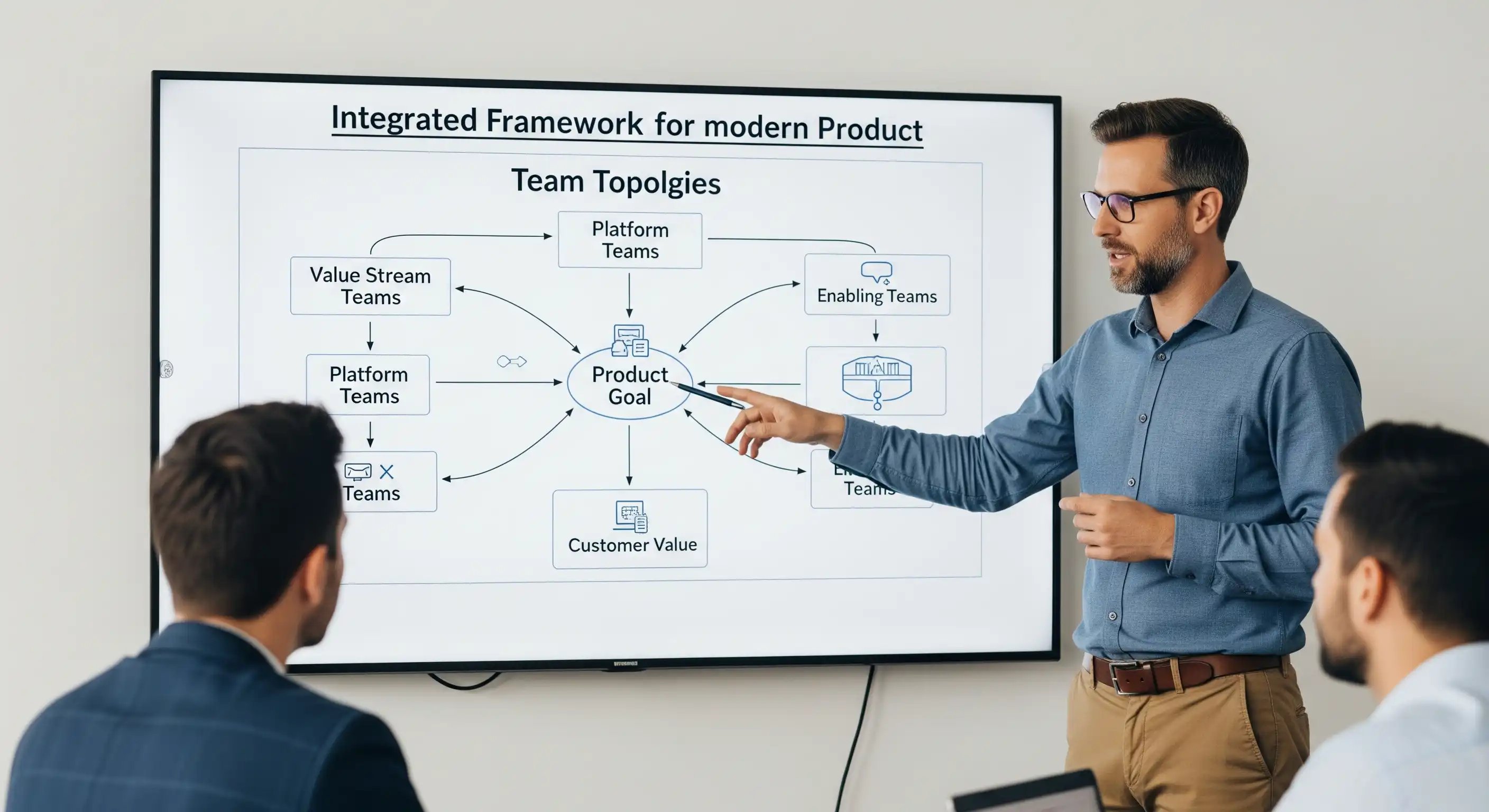
Achieving Peak Performance: The Journey to a Mature Product Organization
Unlock the secrets to transforming your business into a high-performing, Mature Product Organization that consistently delivers exceptional customer value and drives growth. This article explores the essential characteristics of elite product teams and provides a roadmap to navigate the challenges of this critical evolution.
Navigating the Seismic Shift: FDA Finalizes Transition to QMSR, Harmonizing with ISO 13485
The FDA is fundamentally changing its medical device regulations by harmonizing with the global ISO 13485 standard. Discover what the new Quality Management System Regulation (QMSR) means for manufacturers and the critical steps needed to prepare for the February 2, 2026 deadline.
Ready for a Digital Revolution? Deep Dive into the EU's Game-Changing Cyber Resilience Act
Are you ready to understand the future of the connected devices that shape your daily life? We’re not talking about a minor software update; we're talking about a fundamental reshaping of our digital world.
EU's New Life Sciences Strategy: A 'Choose Europe' Game Plan for Innovation
Discover the European Commission's game-changing "Choose Europe" strategy, an ambitious plan to make the EU a global life sciences leader by 2030. Read our full analysis to understand the critical impacts on medical device and pharmaceutical manufacturers and how to align with Europe's accelerated push for innovation.
Audio Deep Dive: Unpacking Europe's Grand Vision for Life Sciences
Listen to our deep dive on the EU's 'Choose Europe for life sciences' strategy. We unpack the ambitious plan to become a global leader in MedTech, biotech, and pharmaceuticals by 2030.
Pharma's Geopolitical Chessboard: Navigating US vs. EU Regulation in 2025
The global pharmaceutical landscape is undergoing seismic shifts, transforming into a geopolitical chessboard due to wildly divergent US "America First" and EU "Pharma Package" regulatory changes. Dive into our latest post and recording to understand these critical shifts, their impact on supply chains and innovation, and what they mean for the future of medicines and global collaboration.
2025 Healthcare & Life Sciences Quality and Regulatory Infographic
Unlock the future of healthcare and life sciences regulation by exploring our comprehensive infographic on Quality and Regulatory in 2025. Understand the critical shifts in global strategies, from AI governance to supply chain demands, to future-proof your approach.
Navigating the Clinical Trial
Discover the intricate journey of medical devices from concept to market and gain a comprehensive understanding of clinical trial pathways. Dive into our detailed guide to equip your team with the essential knowledge for navigating regulatory landscapes and fostering innovation.
Navigating the Clinical Trial Gauntlet
An Interactive Guide to the Medical Device Clinical Trial Pathway for Product & Engineering Leaders. Unlock the future of healthcare and life sciences regulation by exploring our comprehensive infographic on Quality and Regulatory in 2025. Understand the critical shifts in global strategies, from AI governance to supply chain demands, to future-proof your approach.
Navigating the Clinical Trial Infographic
An Interactive Infographic about Medical Device Clinical Trial Pathway for Product & Engineering Leaders.
The End of an Era: How New Approach Methodologies Are Replacing Animal Testing
Explore the FDA's shift away from animal testing and the rise of New Approach Methodologies (NAMs) like computational modeling, organoids, and organs-on-a-chip. Learn what this means for the future of drug and device development.
2025 Regulatory Report: The In-Silico Revolution
An interactive report on the in-silico revolution, exploring the shift away from animal testing towards New Approach Methodologies (NAMs) and computational model validation with ASME V&V 40.
Transforming to a Product Organization: Why Principles Outperform Processes
Transform your organization from process-bound to principle-driven and unlock true innovation and customer delight. Dive into our comprehensive guide to learn how to cultivate empowerment, adaptability, and trust for lasting success.
The Shift from "Old Ways" to Empowered Product Teams
Do your teams consistently ship features that fail to deliver the expected business results? Do your product requirements come from a roadmap handed down by stakeholders, rather than being discovered by an empowered team? Do your best people feel like "mercenaries" just executing tasks, rather than "missionaries" with true ownership over outcomes?
Shifting to a Product Model: The Power of Outcome-Based Roadmaps
Ready to transform your organization from an output-focused factory to an outcome-driven powerhouse? Dive into this article to discover how outcome-based roadmaps and a clear product vision can empower your teams and ignite continuous innovation.
A Technical Review of the EU's Mandatory AI Training Data Disclosure
A deep dive into the EU AI Act's mandatory reporting framework for general-purpose AI models, explaining obligations and compliance.
America's AI Action Plan: What Industry Managers Need to Know
A comprehensive overview of the United States government's AI Action Plan, detailing its pillars, regulatory shifts, and implications for industry leaders.
Is AI a Job-Thief or a Superpower? An Insight into the Ground-Level Effect of Generative AI on Work
Explore real-world data from a Microsoft Bing Copilot study revealing how generative AI is augmenting human capabilities rather than simply automating jobs.
Teams That Solve Problems, Not Backlogs: Empowered Teams From Output to Outcomes in 90 Days
Most product teams burn down backlogs instead of solving customer problems. Learn how empowered teams own outcomes with a practical 90-day plan, metrics, and leadership shifts.
A Regulatory & Quality Expert’s Take on MedTech Europe’s “Simplification of EU Digital Legislation” Position
MedTech Europe has published a position paper urging the EU to “simplify” horizontal digital laws (AI Act, Cybersecurity, Data Act, EHDS) and align them with sectoral frameworks (MDR/IVDR). This reaction summarises the key asks, assesses their feasibility from a QA/RA perspective, and proposes an implementation playbook for manufacturers and policymakers.
From Gridlock to Innovation: How the FDA's CSA Guidance is Reshaping MedTech Software Validation
For years, the medical device industry has operated under the rigid constraints of Computer System Validation (CSV), a framework often criticized for its heavy focus on documentation over actual quality assurance. This approach created a validation gridlock, slowing down the adoption of modern digital tools like eQMS and cloud platforms. But the landscape is changing.
Is Your AI Fair? A Deep Dive into the Hidden Biases of Artificial Intelligence
In a world where AI influences critical decisions from hiring to healthcare, hidden biases can perpetuate unfair outcomes and damage public trust. Leveraging the foundational NIST framework, this article explores the deep-seated causes of bias—from statistical errors to systemic issues—and outlines a practical roadmap for creating more fair and trustworthy AI.
AI Drift: The Silent Risk in Healthcare and Mission-Critical Systems
An AI model is not a static piece of software; its performance can silently degrade as the real world changes, creating significant risks in mission-critical systems. This article breaks down the different types of AI drift and offers a practical guide for quality and regulatory teams on the essential questions they must ask to ensure safety and effectiveness in regulated industries like healthcare.
The MedTech Startup's Compass: Navigating the Regulatory Valley of Death
This post argues that the MedTech "valley of death" is regulatory, not technical, demonstrating how startups fail by treating the FDA submission as a final checklist instead of a continuous trust-building exercise. It outlines a stakeholder-driven philosophy for success, detailing how to build a coherent, evidence-based narrative by embedding clinician and patient input directly into foundational documents like the Design History File, Risk Management File, and Human Factors file.
Engineering the EHDS: A Technical Blueprint for Hospital CIOs
The European Health Data Space (EHDS) Regulation has fundamentally altered the role of hospital IT. We are moving away from a regime of legal compliance—where checkboxes sufficed—to a regime of engineering physics. The regulation mandates specific architectural constraints, most notably the Secure Processing Environment (SPE), which requires technical isolation so rigorous that data "visitation" replaces data "transfer."
The Fair Game: How to Fix AI Bias as the World Changes
When we talk about "AI safety," a very big word is fairness. Basically, we want the AI to stay good even when things change or when it has to make difficult choices. Most AI models learn from the past, but the past contains inequality and unfair rules. So the AI does not just learn the task, it also learns the habits we should be trying to leave behind.
Lego and 3D Printing Together: The Hybrid Prototyping Method That Works
New research from the University of Bristol shows that combining Lego bricks with 3D printing can cut prototype fabrication time by 56% and material costs by 76%. Learn how the three hybrid strategies work and when to apply each one.
Detecting APT Attacks With Sparse Data: A Knowledge Graph Embedding Approach That Works
Most knowledge graph embedding systems fail on APT data because the graphs are structurally sparse. APT-ST-AN addresses this by adding sequential attack relations and adversarial hard negatives, doubling usable training triples and improving link prediction accuracy.
From Submission Wave to Strategic Mandate: Why AI in Drug Development Is Here to Stay
The FDA has already reviewed over 500 drug submissions containing AI components. For US and EU life sciences corporations, this is not a pilot phase — it is the new operating baseline. The disruption has arrived, and it is not leaving.
AI Governance Is Not a Final Layer: Why Shifting It Left Is the Only Strategy That Scales
40% of enterprises admit their AI governance is inadequate. The root cause is always the same: governance treated as a final checkpoint rather than a foundational design principle. For CTOs, board leaders, and regulatory affairs specialists, the time to fix this is before the next AI system ships.
FDA Replaced QSIT: How Risk Management Files Now Drive Device Inspections
FDA replaced QSIT with a new risk-based inspection model under QMSR. Investigators now start with your risk management file — and that changes how you prepare for every inspection.
Not All AI Is the Same: The Hardware That Powers Different Machine Learning Systems
AI is not one monolithic technology — the hardware underneath each model type varies significantly. This article breaks down CPUs, GPUs, NPUs, compute-in-memory, and neuromorphic chips, and explains which workloads each one actually fits.
What AI Cannot Do: The Leadership Skills That Matter More Than Ever
AI is automating the transactional work of leadership. What it cannot do is set direction, build trust, or make judgment calls under uncertainty. This post explores what leadership actually requires in the AI era — and why those capabilities are more valuable now than ever.
How LoRA Makes Custom AI Affordable for Enterprise Backoffice Operations
Fine-tuning a large language model used to mean a six-figure GPU budget and months of compute time. LoRA changes that equation entirely. This post explores how Low-Rank Adaptation enables enterprise teams to build domain-specific AI for backoffice operations — faster, cheaper, and without sacrificing meaningful accuracy.
EU AI Act 2026: What Compliance Officers Must Do Before August
The EU AI Act enters full applicability on 2 August 2026, and several key obligations will bite before that date. For data protection officers, legal counsels, and compliance managers, this is not a distant regulatory exercise. It is a live programme of work with hard deadlines, meaningful fines, and board-level reputational exposure.
The Coordination Tax: Why AI Tools Won't Fix Your Team Size Problem
AI note-takers are making overcrowded meetings more bearable — which is exactly why they are dangerous. The real problem is not information overload, it is the geometric cost of large teams. Here is how to redesign your organization before AI amplifies the damage.
OpenClaw Security: What Enterprise Teams Must Do Before Deploying AI Agents
OpenClaw reached 201,000 GitHub stars in days — and arrived with critical vulnerabilities already being exploited at scale. Before your team deploys it, there are ten hardening steps that separate a manageable risk from an active liability.
FDA's CDS Final Guidance: What AI/ML Medical Device Teams Must Know
FDA's final CDS guidance is now in force — and it applies to LLMs without exception. For teams developing SaMD and AI-enabled clinical software, the four criteria from the 21st Century Cures Act are the governing framework. Here is what the Town Hall Q&A revealed about Criterion 4, alarms, and where most regulatory risk actually concentrates.
AI Did Not Automate Your Jobs — It Revealed They Were Never the Real Work
The Block layoffs made headlines, but the real story is not about automation. AI is eliminating the coordination layer that most organisations are built around — and leaders who wait to feel it will have less room to respond than those who redesign now.
Four Ways AI Agents Fail When the Stakes Are High
A landmark Mount Sinai study on ChatGPT Health revealed that AI agents recommended against emergency care in over half of urgent cases. The four structural failure modes exposed in this research are not specific to healthcare — they are almost certainly present in your enterprise agents right now.
The Axios Attack: Why You Cannot Trust Your npm Dependencies by Default
An attacker used stolen maintainer credentials to publish poisoned Axios versions that installed a Remote Access Trojan on developer machines during npm install. Standard audit tools show nothing after the fact. Here is what happened and what your team must do.
A more practical operating model for technology leaders and engineering managers evaluating enterprise AI systems
Agents It shows why agentic ai is not just a bigger model, it is a system design problem involving autonomy, adaptability, control, and governance
A Technical Review of a Lightweight CNN for Invasive Ductal Carcinoma Detection
How a redesigned CNN improved accuracy and recall for breast histopathology while sharply reducing GPU memory demand.
Traditional OCR vs Generative AI-Assisted OCR: What Changes for Healthcare Operations?
Why document automation is moving from plain text recognition to contextual extraction, workflow integration, and stronger operational governance.
OpenMythos and the Recurrent-Depth Transformer Bet: Why AI Architects Should Pay Attention
A technical but accessible analysis of OpenMythos, recurrent-depth transformers, stable looped language models, and what the latest research means for AI architects.
Google Open-Sources agents-cli: Why Architects and Engineers Should Pay Attention
Google has open-sourced agents-cli, a CLI and skills layer for building, evaluating, and deploying agents on Google Cloud. Here is what that means for architects and engineers.
How AI Will Disrupt and Augment Sales and Marketing Teams in European Automotive Retail
AI is reshaping how European dealerships and online car platforms generate leads, personalise finance conversations, convert service customers, and improve sales productivity.
Why the FDA's Real-Time Trial Signal Changes Clinical Operations Now
The FDA has already validated real-time signals in an AstraZeneca study and is launching an AI-enabled early-phase pilot that could reshape clinical operations, regulatory strategy, and sponsor readiness.
AI Risk Is Not Just ISMS With Better Marketing
AI risk overlaps with ISMS risk, but it also introduces model behaviour, decision harm, bias, drift, and overreliance risks that a traditional ISMS does not fully govern.
Same Boardroom, Different Beast: AI Risk vs ISMS Risk Side by Side
AI risk and ISMS risk overlap, but they differ in risk object, failure mode, evidence, ownership, and control style.
Where They Collide: The Shared Zone Between Cybersecurity and AI Governance
AI risk and cybersecurity risk collide in data, access, prompt injection, supply chain, logging, and incident response.
FDA’s May eSTAR FAQ Update Looks Minor — But Submission Teams Should Not Ignore It
The FDA did not release new eSTAR PDFs on May 1, but its updated FAQ guidance still matters for 510(k), De Novo, PMA, IDE, and Q-Submission teams preparing device filings.
The Risks Security Teams Underestimate in AI
Many of the most serious AI failures are not classic security failures. They emerge through unreliable outputs, hidden bias, weak explainability, human overreliance, and silent model drift.
The Five Layers of Agentic AI: From Models to Operating Model
Agentic AI is not just an agent on top of a model. It is a five-layer stack that runs from AI and deep learning to GenAI, agents, and the operating model required for safe execution at scale.
From Controls to Accountability: Designing a Governance Model That Actually Works
AI risk management fails when responsibility stays vague. A working model requires explicit ownership across security, legal, compliance, data, product, and business leadership.
One Risk Language, Two Lenses: How to Integrate AI Risk Into an Existing ISMS
The goal is not to replace the ISMS. It is to extend enterprise risk discipline so AI-specific harms are governed with the same seriousness, but with the additional methods and evidence AI requires.
Article 61(10) Is Not a Shortcut — It Is a Higher Burden of Justification
A point-of-view response to the Team-NB Article 61(10) position paper: why clinical evaluation based on non-clinical data is not a shortcut, but a narrower and more demanding evidentiary route under the MDR.
Porter’s Five Forces After AI: Why Intent-Driven Organizations Compete Differently
AI does not cancel Porter’s Five Forces. It changes how each force is expressed, and it rewards organizations that push decision-making closer to the work through an intent-driven model.
Memory Sparse Attention: Why 100M-Token Memory Could Change Agentic AI
A technical review of the MSA paper and why scalable intrinsic memory, not just bigger context windows, may unlock the next leap for agents and generative AI.
The EU AI Act Delay Changes More Than the Calendar for MedTech and Other High-Risk AI Teams
The stalled EU AI Act trilogue negotiations may push key high-risk AI obligations from August 2026 to December 2027. For MedTech and other Annex I product-regulated teams, that changes timing, not accountability.
The Silent Revolution Behind HTTP 402: Why x402 Could Rewire Agentic Commerce and Europe’s Payment Future
HTTP 402 was reserved in 1991 and then mostly ignored. x402 is bringing it back as a machine-native payment rail for APIs, agents, and programmable settlement—and Europe should pay attention.
LoRA Is Still the Smartest Way to Train Open Models for Real Domain Work
LoRA is still the cleanest way to train open models for domain-specific work because it cuts compute, shortens iteration cycles, and lets teams ship specialised adapters instead of whole new models.
FDA’s Elsa 4.0 and HALO Signal a Bigger Shift in Regulatory Operations
The FDA’s Elsa 4.0 launch and HALO data consolidation matter less as isolated AI features and more as a sign that regulatory operations are being rebuilt around unified data and AI-enabled workflows.
Navigating the MedTech Shift from Computer System Validation to Computer Software Assurance
A written introduction to the shift from Computer System Validation to Computer Software Assurance, focused on the validation paradox, the limits of traditional CSV, and the FDA’s risk-based CSA framework.